Artículos SCI
2017
2017
Materiales de Diseño para la Energía y Medioambiente
Cellulose-polyhydroxylated fatty acid ester-based bioplastics with tuning properties: Acylation via a mixed anhydride system
Heredia-Guerrero, JA; Goldoni, L; Benitez, JJ; Davis, A; Ceseracciu, L; Cingolani, R; Bayer, IS; Heinze, T; Koschella, A; Heredia, A; Athanassiou, ACarbohydrate Polymers, 173 (2017) 312-320
DOI: 10.1016/j.carbpol.2017.05.068
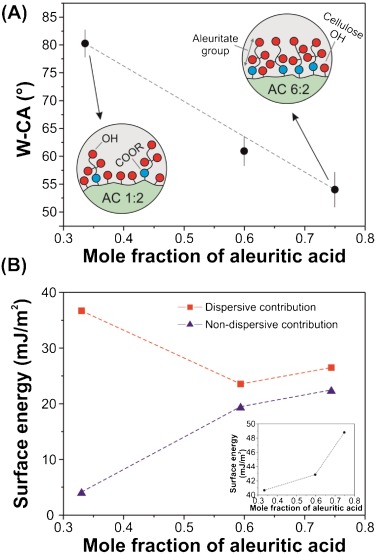
Abstract
The synthesis of microcrystalline cellulose (MCC) and 9,10,16-hydroxyhexadecanoic (aleuritic) acid ester-based bioplastics was investigated through acylation in a mixed anhydride (trifluoroacetic acid (TFA)/trifluoroacetic acid anhydride (TFAA)), chloroform co-solvent system. The effects of chemical interactions and the molar ratio of aleuritic acid to the anhydroglucose unit (AGU) of cellulose were investigated. The degree of substitution (DS) of new polymers were characterized by two-dimensional solution-state NMR and ranged from 0.51 to 2.60. The chemical analysis by attenuated total reflection Fourier transform infrared spectroscopy (ATR-FTIR) confirmed the presence of aleuritate groups in the structure induces the formation of new H-bond networks. The tensile analysis and the contact angle measurement confirmed the ductile behavior and the hydrophobicity of the prepared bioplastics. By increasing the aleuritate amounts, the glass transition temperature decreased and the solubility of bioplastic films in most common solvents was improved. Furthermore, this new polymer exhibits similar properties compared to commercial cellulose derivatives.

- Citation Indexes: 36
- Patent Family Citations: 3
- Readers: 78
- News Mentions: 1
Octubre, 2017 · DOI: 10.1016/j.carbpol.2017.05.068
Química de Superficies y Catálisis
Synthesis and characterization of Rh/MnO2-CeO2/Al2O3 catalysts for CO-PrOx reaction
Martinez, TLM; Laguna, OH; Lopez-Cartes, C; Centeno, MAMolecular Catalysis, 440 (2017) 9-18
DOI: 10.1016/j.mcat.2017.06.018
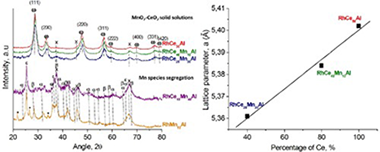
Abstract
Rh/MnO2-CeO2/Al2O3 catalysts with different manganese-to-ceria ratios have been synthesized, characterized and tested in CO-PrOx reaction. The physicochemical properties of the solids were studied by XRD, Raman spectroscopy, BET surface area, H-2-TPR, TGA-DTG and TEM. The differences observed in the textural, structural and redox properties were related to the Mn-to-ceria ratio of the samples. The segregation of Mn species was observed at high Mn-to-Ce ratios. In opposite way, MnO2-CeO2 solid solutions were obtained at low Mn to Ce ones. In this last case, the physicochemical properties of the solids were favored by the intimate Rh-Ce-Mn contact. The effect of the Mn-Ce presence on Rh catalysts which promotes the catalytic behavior towards selective CO oxidation was observed to be better at low temperatures. At higher temperatures, Mn species promote the Reverse Water Gas Shift reaction, whilst ceria promotes the H-2 oxidation in the whole range of working temperatures.
Octubre, 2017 · DOI: 10.1016/j.mcat.2017.06.018
Materiales y Procesos Catalíticos de Interés Ambiental y Energético - Materiales Nanoestructurados y Microestructura
Towards Extending Solar Cell Lifetimes: Addition of a Fluorous Cation to Triple Cation-Based Perovskite Films
Salado, M; Fernandez, MA; Holgado, JP; Kazim, S; Nazeeruddin, MK; Dyson, PJ; Ahmad, SChemsuschem, 10 (2017) 3846-3853
DOI: 10.1002/cssc.201700797
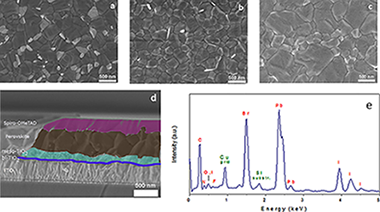
Abstract
Organohalide perovskites have emerged as highly promising replacements for thin-film solar cells. However, their poor stability under ambient conditions remains problematic, hindering commercial exploitation. The addition of a fluorous-functionalized imidazolium cation during the preparation of a highly stable cesium-based mixed perovskite material Cs-0.05(MA(0.15)FA(0.85))(0.95)Pb(I0.85Br0.15)(3) (MA= methylammonium; FA= formamidinium) has been shown to influence its stability. The resulting materials, which vary according to the amount of the fluorous-functionalized imidazolium cation present during fabrication, display a prolonged tolerance to atmospheric humidity (> 100 days) along with power conversion efficiencies exceeding 16%. This work provides a general route that can be implemented in a variety of perovskites and highlights a promising way to increase perovskite solar cell stability.
Octubre, 2017 · DOI: 10.1002/cssc.201700797
Reactividad de Sólidos
Dense graphene nanoplatelet/yttria tetragonal zirconia composites: Processing, hardness and electrical conductivity
Gallardo-Lopez, A; Marquez-Abril, I; Morales-Rodriguez, A; Munoz, A; Poyato, RCeramics International, 43 (2017) 11743-11752
DOI: 10.1016/j.ceramint.2017.06.007

Abstract
Yttria tetragonal zirconia ceramic composites with 1, 2.5, 5 and 10 vol% nominal contents of graphene nanoplatelets (GNPs) were fabricated and characterized. First, the GNP dispersion in isopropanol was optimized to de-agglomerate the GNPs without damaging their structure. Then, submicrometric fully dense composites were obtained via spark plasma sintering (SPS) at 1250 degrees C with a 5 min holding time. The processing routine produced a nearly homogeneous GNP dispersion in the ceramic matrix, and the GNPs preferential orientation was perpendicular to the sintering compression axis. A ceramic grain refinement due to the GNPs was also detected. The Vickers hardness measured on the plane perpendicular to the sintering compression axis (basal plane) was lower than on the cross sections. This anisotropy increased with the increasing GNP content, while the average hardness decreased. The electrical conductivity was also highly anisotropic, up to seven times higher for the basal planes. The electrical percolation threshold for these composites was estimated to be between 2.2 and 4.4 vol% of the GNP measured content.
Octubre, 2017 · DOI: 10.1016/j.ceramint.2017.06.007
Nanotecnología en Superficies y Plasma
Vapor and liquid optical monitoring with sculptured Bragg microcavities
Oliva-Ramirez, M; Gil-Rostra, J; Lopez-Santos, MC; Gonzalez-Elipe, AR; Yubero, FJournal of Nanophotonics, 11 (2017) 046009
DOI: 10.1117/1.JNP.11.046009
Abstract
Sculptured porous Bragg microcavities (BMs) formed by the successive stacking of columnar SiO2 and TiO2 thin films with a zig-zag columnar microstructure are prepared by glancing angle deposition. These BMs act as wavelength-dependent optical retarders. This optical behavior is attributed to a self-structuration of the stacked layers involving the lateral association of nanocolumns in the direction perpendicular to the main flux of particles during the multilayer film growth, as observed by focused ion beam scanning electron microscopy. The retardance of these optically active BMs can be modulated by dynamic infiltration of their open porosity with vapors, liquids, or solutions with different refractive indices. The tunable birefringence of these nanostructured photonic systems has been successfully simulated with a simple model that assumes that each layer within the BMs stack has uniaxial birefringence. The sculptured BMs have been incorporated as microfluidic chips for optical transduction for label-free vapor and liquid sensing. Several examples of the detection performance of these chips, working either in reflection or transmission configuration, for the optical monitoring of vapor and liquids of different refractive indices and aqueous solutions of glucose flowing through the microfluidic chips are described.
Octubre, 2017 · DOI: 10.1117/1.JNP.11.046009
Tribología y Protección de Superficies - Materiales Coloidales
HoF3 and DyF3 Nanoparticles as Contrast Agents for High-Field Magnetic Resonance Imaging
Gonzalez-Mancebo, Daniel; Becerro, Ana I.; Rojas, T. Cristina; Garcia-Martin, Maria L.; de la Fuente, Jesus M.; Ocana, ManuelParticle & particle systems characterization, 34 (2017) art. 1700116
DOI: 10.1002/ppsc.201700116
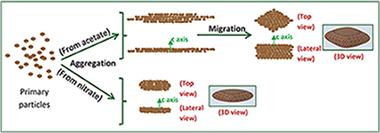
Abstract
Clinical contrast agents (CAs) currently used in magnetic resonance imaging (MRI) at low fields are less effective at high magnetic fields. The development of new CAs is mandatory to improve diagnostic capabilities of the new generation of high field MRI scanners. The purpose of this study is to synthesize uniform, water dispersible LnF3 (Ln = Ho, Dy) nanoparticles (NPs) and to evaluate their relaxivity at high magnetic field (9.4 T) as a function of size and composition. Two different types of HoF3 NPs are obtained by homogeneous precipitation in ethylene glycol at 120 °C. The use of holmium acetate as holmium precursor leads to rhombus-like nanoparticles, while smaller, ellipsoid-like nanoparticles are obtained when nitrate is used as the holmium salt. To explain this behavior, the mechanism of formation of both kinds of particles is analyzed in detail. Likewise, rhombus-like DyF3 nanoparticles are prepared following the same method as for the rhombus-like HoF3 nanoparticles. We have found, to the best of knowledge, the highest transverse relaxivity values at 9.4 T described in the literature for this kind of CAs. Finally, the LnF3 NPs have shown negligible cytotoxicity for C6 rat glioma cells for concentrations up to 0.1 mg mL−1.
Octubre, 2017 · DOI: 10.1002/ppsc.201700116
Reactividad de Sólidos
Multicycle activity of natural CaCO3 minerals for thermochemical energy storage in Concentrated Solar Power plants
Benitez-Guerrero, M; Valverde, JM; Sanchez-Jimenez, PE; Perejon, A; Perez-Maqueda, LASolar Energy, 153 (2017) 188-199
DOI: 10.1016/j.solener.2017.05.068
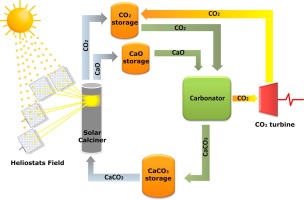
Abstract
Thermochemical energy storage in Concentrated Solar Power plants by means of the Calcium-Looping process is a promising novel technology that would allow for a higher share of renewables. A main benefit of this technology is the use of widely available, non-toxic and environmentally friendly calcium carbonate minerals as raw materials to store energy. Efficient integration of the Calcium-Looping process into Concentrated Solar Power plants involves the endothermic calcination of CaCO3 in the solar receiver while the exothermic carbonation of CaO is carried out at high temperature under high CO2 partial pressure. The heat released by this reaction is carried out by the excess CO2 and employed for power generation by means of a closed CO2 cycle. This work explores the multicycle Calcium-Looping performance of naturally occurring CaCO3 minerals such as limestone, chalk and marble for thermochemical energy storage in Concentrated Solar Power plants. Despite their similar composition (almost pure CaCO3), these minerals exhibit a significant difference in their Calcium-Looping multicycle activity, which may be attributed to differences in particle size and microstructure. Pore plugging at the Calcium-Looping conditions for thermochemical energy storage tested in our work is a main limiting mechanism on the multicycle CaO carbonation activity.
Septiembre, 2017 · DOI: 10.1016/j.solener.2017.05.068
Nanotecnología en Superficies y Plasma
High performance novel gadolinium doped ceria/yttria stabilized zirconia/nickel layered and hybrid thin film anodes for application in solid oxide fuel cells
Garcia-Garcia, FJ; Beltran, AM; Yubero, E; Gonzalez-Elipe, AR; Lambert, RMJournal of Power Sources, 363 (2017) 251-259
DOI: 10.1016/j.jpowsour.2017.07.085

Abstract
Magnetron sputtering under oblique angle deposition was used to produce Ni-containing ultra thin film anodes comprising alternating layers of,gadolinium doped ceria (GDC) and yttria stabilized zirconia (YSZ) of either 200 nm or 1000 nm thickness. The evolution of film structure from initial deposition, through calcination and final reduction was examined by XRD, SEM, TEM and TOF-SIMS. After subsequent fuel cell usage, the porous columnar architecture of the two-component layered thin film anodes was maintained and their resistance to delamination from the underlying YSZ electrolyte was superior to that of corresponding single component Ni-YSZ and Ni-GDC thin films. Moreover, the fuel cell performance of the 200 nm layered anodes compared favorably with conventional commercially available thick anodes. The observed dependence of fuel cell performance on individual layer thicknesses prompted study of equivalent but more easily fabricated hybrid anodes consisting of simultaneously deposited Ni-GDC and Ni-YSZ, which procedure resulted in exceptionally intimate mixing and interaction of the components. The hybrids exhibited very unusual and favorable I-V characteristics, along with exceptionally high power densities at high currents. Their discovery is the principal contribution of the present work.
Septiembre, 2017 · DOI: 10.1016/j.jpowsour.2017.07.085
Materiales Coloidales
Europium-doped NaGd(WO4)(2) nanophosphors: synthesis, luminescence and their coating with fluorescein for pH sensing
Laguna, M; Escudero, A; Nuñez, NO; Becerro, AI; Ocaña, MDalton Transactions, 46 (2017) 11575-11583
DOI: 10.1039/c7dt01986f
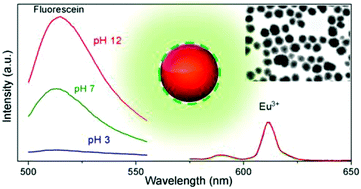
Abstract
Uniform Eu-doped NaGd(WO4)(2) nanophosphors with a spherical shape have been synthesized for the first time by using a wet chemistry method based on a homogeneous precipitation process at low temperature (120 degrees C) in ethylene glycol/water mixtures. The obtained nanoparticles crystallized into the tetragonal structure and presented polycrystalline character. The europium content in such phosphors has been optimized through the analysis of the luminescence dynamics (lifetime measurements). By coating the Eu3+-doped wolframate based nanoparticles with fluorescein through a layer-by-layer (LbL) approach, a wide range (4-10) ratiometric pH-sensitive sensor has been developed, which uses the pH insensitive emission of Eu3+ as a reference.

Septiembre, 2017 · DOI: 10.1039/c7dt01986f
Nanotecnología en Superficies y Plasma
In Vitro and in Vivo Study of Poly(Lactic-co-Glycolic) (PLGA) Membranes Treated with Oxygen Plasma and Coated with Nanostructured Hydroxyapatite Ultrathin Films for Guided Bone Regeneration Processes
Torres-Lagares, D; Castellanos-Cosano, L; Serrera-Figallo, MA; Garcia-Garcia, FJ; Lopez-Santos, C; Barranco, A; Elipe, ARG; Rivera-Jimenez, C; Gutierrez-Perez, JLPolymers, 9 (2017) art. 410
DOI: 10.3390/polym9090410
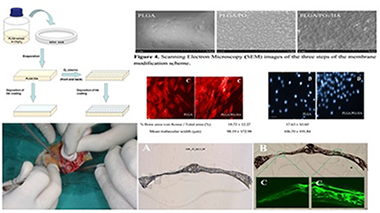
Abstract
The novelty of this study is the addition of an ultrathin layer of nanostructured hydroxyapatite (HA) on oxygen plasmamodified poly(lactic-co-glycolic) (PLGA) membranes (PO2) in order to evaluate the efficiency of this novel material in bone regeneration. Methods: Two groups of regenerative membranes were prepared: PLGA (control) and PLGA/PO2/HA (experimental). These membranes were subjected to cell cultures and then used to cover bone defects prepared on the skulls of eight experimental rabbits. Results: Cell morphology and adhesion of the osteoblasts to the membranes showed that the osteoblasts bound to PLGA were smaller and with a lower number of adhered cells than the osteoblasts bound to the PLGA/PO2/HA membrane (p < 0.05). The PLGA/PO2/HA membrane had a higher percentage of viable cells bound than the control membrane (p < 0.05). Both micro-CT and histological evaluation confirmed that PLGA/PO2/HA membranes enhance bone regeneration. A statistically significant difference in the percentage of osteoid area in relation to the total area between both groups was found. Conclusions: The incorporation of nanometric layers of nanostructured HA into PLGA membranes modified with PO2 might be considered for the regeneration of bone defects. PLGA/PO2/HA membranes promote higher osteosynthetic activity, new bone formation, and mineralisation than the PLGA control group.
Septiembre, 2017 · DOI: 10.3390/polym9090410
Materiales Coloidales
Rare earth based nanostructured materials: synthesis, functionalization, properties and bioimaging and biosensing applications
Escudero, Alberto; Becerro, Ana I.; Carrillo-Carrion, Carolina; Nunez, Nuria O.; Zyuzin, Mikhail V.; Laguna, Mariano; Gonzalez-Mancebo, Daniel; Ocana, Manuel; Parak, Wolfgang J.Nanophotonics, 6 (2017) 881-921
DOI: 10.1515/nanoph-2017-0007
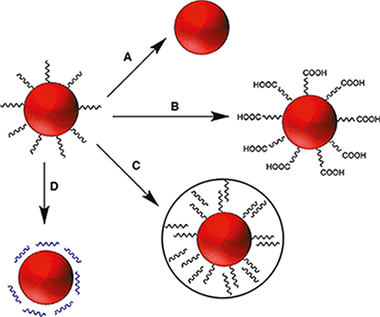
Abstract
Rare earth based nanostructures constitute a type of functional materials widely used and studied in the recent literature. The purpose of this review is to provide a general and comprehensive overview of the current state of the art, with special focus on the commonly employed synthesis methods and functionalization strategies of rare earth based nanoparticles and on their different bioimaging and biosensing applications. The luminescent (including downconversion, upconversion and permanent luminescence) and magnetic properties of rare earth based nanoparticles, as well as their ability to absorb X-rays, will also be explained and connected with their luminescent, magnetic resonance and X-ray computed tomography bioimaging applications, respectively. This review is not only restricted to nanoparticles, and recent advances reported for in other nanostructures containing rare earths, such as metal organic frameworks and lanthanide complexes conjugated with biological structures, will also be commented on.
Septiembre, 2017 · DOI: 10.1515/nanoph-2017-0007
Materiales de Diseño para la Energía y Medioambiente
High-temperature thermal conductivity of biomorphic SiC/Si ceramics
Ramirez-Rico, J.; Singh, M.; Zhu, D.; Martinez-Fernandez, J.Journal of Materials Science, 52 (2017) 10038-10046
DOI: 10.1007/s10853-017-1199-y
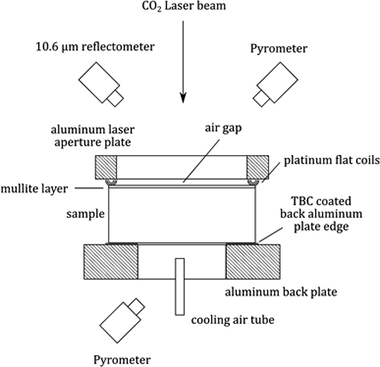
Abstract
Thermal conductivity of biomorphic SiC/Si, a silicon carbide + silicon containing two phase material, was evaluated using the laser steady-state heat flux method. These materials were processed via silicon melt infiltration of wood-derived carbon scaffolds. In this approach, heat flux was measured through the thickness when one side of the specimen was heated with a 10.6-A mu m CO2 laser. A thin mullite layer was applied to the heated surface to ensure absorption and minimize reflection losses, as well as to ensure a consistent emissivity to facilitate radiative loss corrections. The influence of the mullite layer was accounted for in the thermal conductivity calculations. The effect of microstructure and composition (inherited from the wood carbonaceous performs) on measured conductivity was evaluated. To establish a baseline for comparison, a dense, commercially available sintered SiC ceramic was also evaluated. It was observed that at a given temperature, thermal conductivity falls between that of single-crystal silicon and fine-grained polycrystalline SiC and can be rationalized in terms of the SiC volume fraction in biomorphic SiC/Si material.
Septiembre, 2017 · DOI: 10.1007/s10853-017-1199-y
Nanotecnología en Superficies y Plasma
Enhanced green fluorescent protein in optofluidic Fabry-Perot microcavity to detect laser induced temperature changes in a bacterial culture
Lahoz, F; Martin, IR; Walo, D; Freire, R; Gil-Rostra, J; Yubero, F; Gonzalez-Elipe, ARApplied Physics Letters, 111 (2017) 111103
DOI: 10.1063/1.4990870

Abstract
Thermal therapy using laser sources can be used in combination with other cancer therapies to eliminate tumors. However, high precision temperature control is required to avoid damage in healthy surrounding tissues. Therefore, in order to detect laser induced temperature changes, we have used the fluorescence signal of the enhanced Green Fluorescent Protein (eGFP) over-expressed in an E. coli bacterial culture. For that purpose, the bacteria expressing eGFP are injected in a Fabry-Perot (FP) optofluidic planar microcavity. In order to locally heat the bacterial culture, external infrared or ultraviolet lasers were used. Shifts in the wavelengths of the resonant FP modes are used to determine the temperature increase as a function of the heating laser pump power. Laser induced local temperature increments up to 6-7 degrees C were measured. These results show a relatively easy way to measure laser induced local temperature changes using a FP microcavity and using eGFP as a molecular probe instead of external nanoparticles, which could damage/alter the cell. Therefore, we believe that this approach can be of interest for the study of thermal effects in laser induced thermal therapies.
Septiembre, 2017 · DOI: 10.1063/1.4990870
Reactividad de Sólidos
Influence of milling parameters on the solid-gas synthesis of TiCxN1-x by mechanically induced self-sustaining reaction
Chicardi, E; Gotor, FJ; Alcala, MD; Cordoba, JMPowder Technology, 319 (2017) 12-18
DOI: 10.1016/j.powtec.2017.06.035
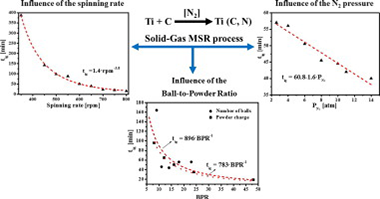
Abstract
The synthesis of a titanium carbonitride solid solution (TiCxN1 − x) performed in a high-energy planetary mill by a solid-gas Mechanically induced Self-sustaining Reaction (MSR) was used to study the influence of a full set of experimental milling parameters on the ignition time (tig) as a measure of the mechanical dose rate provided by the mill. The highly exothermic Ti-C-N mixture was selected to ensure no competitiveness between MSR and diffusion-controlled routes under the milling conditions employed. The results showed that the dependence of tig on the spinning rate followed a potential function, with a potential factor higher than the value of 3 that would be obtained if a perfect collision model is assumed. The scalability of milling processes performed using planetary mills was confirmed. The results suggested that to define a milling experiment, it is necessary to provide not only the Ball-to-Powder mass Ratio (BPR) and spinning rate values, as is usually performed, but also the full set of milling parameters including the nature of the milling media (vial and balls), the number and size of balls, the mass of the powder charge, the pressure of the reactive gas and even the volume of the vial.
Septiembre, 2017 · DOI: 10.1016/j.powtec.2017.06.035
Propiedades mecánicas, modelización y caracterización de cerámicos avanzados
Carbon nanofibers replacing graphene oxide in ceramic composites as a reinforcing-phase: Is it feasible?
Cano-Crespo, Rafael; Malmal Moshtaghioun, Bibi; Gomez-Garcia, Diego; Dominguez-Rodriguez, Arturo; Moreno, RodrigoJournal of the European Ceramic Society, 37 (2017) 3791-3796
DOI: 10.1016/j.jeurceramsoc.2017.03.027

Abstract
In recent years, the interest of graphene and graphene-oxide has increased extraordinarily due to the outstanding properties concurring in this material. In ceramic science, the possibility of combining excellent electrical conductivities together with an enhancement of mechanical properties has motivated the research in fabrication of graphene oxide-reinforced ceramic composites despite the intrinsic difficulties for sintering. In this work a comparison is made between graphene oxide-reinforced alumina composites and carbon nanofiber-reinforced alumina ones. It will be concluded that the improvement of mechanical properties is scarce, if any. Since carbon nanofibers have also a good electrical conductivity their importance for future applications as a replacement of more sophisticated but expensive graphene-based ceramic composites will be stressed.
Septiembre, 2017 · DOI: 10.1016/j.jeurceramsoc.2017.03.027
Reactividad de Sólidos
Simultaneous adsorption and photocatalytic behavior of hybrid mesoporous ZnS-SiO2 nanocomposite
Emrooz, HBM; Gotor, FJMaterials Research Express, 4 (2017) art. 085037
DOI: 10.1088/2053-1591/aa85cf
Abstract
Mesoporous ZnS-SiO2 nanocomposite was synthesized with a facile process. At first a large pore volume (1.86 cm(3).g(-1)), moderate pore size (about 12.8 nm) and moderate surface area (586 m(2).g(-1)) mesoporous SiO2 was synthesized in an acidic PH using cationic surfactant. ZnS nanoparticles were infiltrated in the porosities of the synthesized SiO2, with a room temperature post grafting method. The synthesized particles have been characterized with transmission electron microscopy (TEM), x-ray diffraction (XRD), x-ray photoelectron spectroscopy (XPS), Brunauer-Emmett-Teller (BET), furrier transformation infrared spectroscopy (FTIR) and diffuse reflectance spectroscopy (DRS). Results confirm the mesoporous characteristics of ZnS-SiO2 nanocomposite with specific surface area as high as 248 m(2).g(-1), pore volume of 0.89 cm(3).g(-1) and average pore diameter of about 14.4 nm. Simultaneous adsorption-photocatalytic behavior of this hybrid mesoporous nanocomposite for degradation of methylene blue (MB) was investigated. The efficiency of this material was compared with that of mesoporous SiO2 and also lone ZnS nanoparticles. Results showed that by using ultraviolet irradiation, ZnS-SiO2 can degrade greater than 85% of MB only after 3 min. The case for lone ZnS is less than 5% after 30 min. Degradation mechanism of MB using ZnS-SiO2 and ultraviolet radiation was explained with simultaneous adsorption and photocatalytic phenomena. Ultraviolet irradiation can degrade adsorbed MB of mesoporous SiO2 which can prevent it from saturation.
Agosto, 2017 · DOI: 10.1088/2053-1591/aa85cf
Nanotecnología en Superficies y Plasma
Optical properties and electronic transitions of zinc oxide, ferric oxide, cerium oxide, and samarium oxide in the ultraviolet and extreme ultraviolet
Pauly, N; Yubero, F; Espinos, JP; Tougaard, SApplied Optics, 56 (2017) 6611-6621
DOI: 10.1364/AO.56.006611
Abstract
Optical properties and electronic transitions of four oxides, namely zinc oxide, ferric oxide, cerium oxide, and samarium oxide, are determined in the ultraviolet and extreme ultraviolet by reflection electron energy loss spectroscopy using primary electron energies in the range 0.3 - 2.0 keV. This technique allows the evaluation of the optical response in these ultraviolet spectral regions of a thin layer of material, and the analysis is straightforward. It is performed within the dielectric response theory by means of the QUEELS-epsilon(k,omega)-REELS software developed by Tougaard and Yubero [Surf. Interface Anal. 36, 824 ( 2004)]. The method consists basically in the fitting of experimentally determined single-scattering electron energy loss cross sections with a parametric energy loss function of the corresponding material, to the one calculated within a dielectric response formalism. The obtained refractive index and extinction coefficients, as well as the identified electronic transitions are compared, when available, with previously published results.
Agosto, 2017 · DOI: 10.1364/AO.56.006611
Materiales de Diseño para la Energía y Medioambiente
Cutin from agro-waste as a raw material for the production of bioplastics
Heredia-Guerrero, JA; Heredia, A; Dominguez, E; Cingolani, R; Bayer, IS; Athanassiou, A; Benitez, JJJournal of Experimental Botany, 68 (2017) 5401-5410
DOI: 10.1093/jxb/erx272
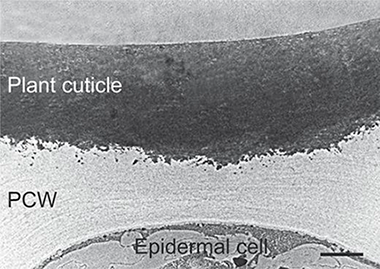
Abstract
Cutin is the main component of plant cuticles constituting the framework that supports the rest of the cuticle components. This biopolymer is composed of esterified bi- and trifunctional fatty acids. Despite its ubiquity in terrestrial plants, it has been underutilized as raw material due to its insolubility and lack of melting point. However, in recent years, a few technologies have been developed to obtain cutin monomers from several agro-wastes at an industrial scale. This review is focused on the description of cutin properties, biodegradability, chemical composition, processability, abundance, and the state of art of the fabrication of cutin-based materials in order to evaluate whether this biopolymer can be considered a source for the production of renewable materials.

- Citation Indexes: 77
- Patent Family Citations: 1
- Policy Citations: 2
- Readers: 240
Agosto, 2017 · DOI: 10.1093/jxb/erx272
Nanotecnología en Superficies y Plasma - Materiales Nanoestructurados y Microestructura
The role of cobalt hydroxide in deactivation of thin film Co-based catalysts for sodium borohydride hydrolysis
Paladini, M; Arzac, GM; Godinho, V; Hufschmidt, D; de Haro, MCJ; Beltran, AM; Fernandez, AApplied Catalysis B-Environmental, 210 (2017) 342-351
DOI: 10.1016/j.apcatb.2017.04.005
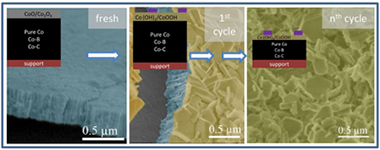
Abstract
Deactivation of a Co catalyst prepared as thin film by magnetron sputtering was studied for the sodium borohydride (SB) hydrolysis reaction under different conditions. Under high SB concentration in single run experiments, the formation of a B-O passivating layer was observed after 1.5 and 24 h use. This layer was not responsible for the catalyst deactivation. Instead, a peeling-off mechanism produced the loss of cobalt. This peeling-off mechanism was further studied in cycling experiments (14 cycles) under low SB concentrations. Ex-situ study of catalyst surface after use and solid reaction products (precipitates) was performed by X-Ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM) and scanning transmission electron microscopy (STEM). The presence of cobalt hydroxide and oxyhydroxide was detected as major components on the catalyst surface after use and as precipitates in the supernatant solutions after washing. Cobalt borate, cobalt carbonate and oxycarbonate were also formed but in lesser amounts. These oxidized cobalt species were formed and further detached from the catalyst at the end of the reaction and/or during catalyst washing by decomposition of the unstable in-situ formed cobalt boride. Leaching of cobalt soluble species was negligible. Thin film mechanical detachment was also found but in a smaller extent. To study the influence of catalyst composition on deactivation processes, cycling experiments were performed with Co-B and Co-C catalysts, also prepared as thin films. We found that the deactivation mechanism proposed by us for the pure Co catalyst also occurred for a different pure Co (prepared at higher pressure) and the Co-B and Co-C samples in our experimental conditions.
Agosto, 2017 · DOI: 10.1016/j.apcatb.2017.04.005
Materiales Coloidales
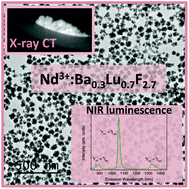
Crystal structure, NIR luminescence and X-ray computed tomography of Nd3+:Ba0.3Lu0.7F2.7 nanospheres
Gonzalez-Mancebo, D; Becerro, AI; Cantelar, E; Cusso, F; Briat, A; Boyer, D; Ocana, MDalton Transactions, 46 (2017) 6580-6587
DOI: 10.1039/c7dt00453b
Abstract
Uniform, hydrophilic 50 nm diameter Nd3+-doped Ba0.3Lu0.7F2.7 nanospheres are synthesized at 120 degrees C using a singular one-pot method based on the use of ethylene glycol as solvent, in the absence of any additive. The composition and crystal structure of the undoped material are analyzed in detail using ICP and XRD, which reveals a BaF2 cubic crystal structure that is able to incorporate 70 mol% of Lu ions. This finding contrasts with the reported phase diagram of the system, where the maximum solubility is around 30 mol% Lu. XRD proves as well that the Ba0.3Lu0.7F2.7 structure is able to incorporate Nd3+ ions up to, at least 10 mol%, without altering the uniform particles morphology. The Nd-doped particles exhibit near-infrared luminescence when excited at 810 nm. The maximum emission intensity with the minimum concentration quenching effect is obtained at 1.5% Nd doping level. X-ray computed tomography experiments are carried out on powder samples of the latter composition. The sample significantly absorbs X-ray photons, thus demonstrating that the Nd3+-doped Ba0.3Lu0.7F2.7 nanospheres are good candidates as contrast agents in computed tomography.
Agosto, 2017 · DOI: 10.1039/c7dt00453b
Materiales de Diseño para la Energía y Medioambiente
New insights into surface-functionalized swelling high charged micas: Their adsorption performance for non-ionic organic pollutants
Pazos, MC; Castro, MA; Cota, A; Osuna, FJ; Pavon, E; Alba, MDJournal of Industrial and Engineering Chemistry, 52 (2017) 179-186
DOI: 10.1016/j.jiec.2017.03.042

Abstract
The major components of the wastewater from the petroleum refineries are benzene, toluene and phenol and one of the techniques applied to the treatment of effluents is sorption using organo-functionalized clay. The materials exploited in the present study are a family of surface-functionalized synthetic micas and their sorption capacities for non-ionic organic pollutants are analyzed. The organo-functionalization of their surface provides them the capacity to sorb effectively non-ionic pollutants in the interface. Their adsorption performance is a function of the alkylamonium properties such as the chain length, the mass fraction and the organization of the organic cation in the interlayer space of the micas.
Agosto, 2017 · DOI: 10.1016/j.jiec.2017.03.042
Reactividad de Sólidos
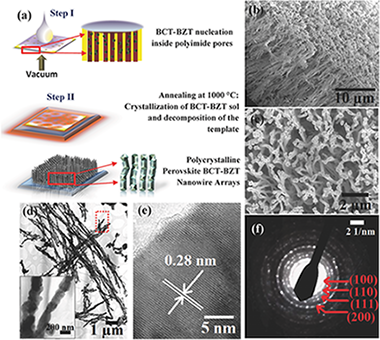
Lead-Free Polycrystalline Ferroelectric Nanowires with Enhanced Curie Temperature
Datta, Anuja; Sanchez-Jimenez, Pedro E.; Al Orabi, Rabih Al Rahal; Calahorra, Yonatan; Ou, Canlin; Sahonta, Suman-Lata; Fornari, Marco; Kar-Narayan, SohiniAdvanced Functional Materials, 27 (2017) 1701169
DOI: 10.1002/adfm.201701169
Abstract
Ferroelectrics are important technological materials with wide-ranging applications in electronics, communication, health, and energy. While lead-based ferroelectrics have remained the predominant mainstay of industry for decades, environmentally friendly lead-free alternatives are limited due to relatively low Curie temperatures (T-C) and/or high cost in many cases. Efforts have been made to enhance T-C through strain engineering, often involving energy-intensive and expensive fabrication of thin epitaxial films on lattice-mismatched substrates. Here, a relatively simple and scalable sol-gel synthesis route to fabricate polycrystalline (Ba0.85Ca0.15)(Zr0.1Ti0.9)O-3 nanowires within porous templates is presented, with an observed enhancement of T-C up to similar to 300 degrees C as compared to similar to 90 degrees C in the bulk. By combining experiments and theoretical calculations, this effect is attributed to the volume reduction in the template-grown nanowires that modifies the balance between different structural instabilities. The results offer a cost-effective solution-based approach for strain-tuning in a promising lead-free ferroelectric system, thus widening their current applicability.
Agosto, 2017 · DOI: 10.1002/adfm.201701169
Materiales de Diseño para la Energía y Medioambiente
Biomorphic ceramics from wood-derived precursors
Ramirez-Rico, J.; Martinez-Fernandez, J.; Singh, M.International Materials Reviews, 62 (2017) Issue 8
DOI: 10.1080/09506608.2017.1354429

Abstract
Materials development is driven by microstructural complexity and, in many cases, inspired by biological systems such as bones, shells and wood. In one approach, one selects the main microstructural features responsible for improved properties and design processes to obtain materials with such microstructures (continuous-fibre-reinforced ceramics, porous ceramics, fibrous ceramic monoliths, etc.). In a different approach, it is possible to use natural materials directly as microstructural templates. Biomorphic ceramics are produced from natural and renewable resources (wood or wood-derived products). A wide variety of SiC-based ceramics can be fabricated by infiltration of silicon or silicon alloys into cellulose-derived carbonaceous templates, providing a low-cost route to advanced ceramic materials with near-net shape potential and amenable to rapid prototyping. These materials have tailorable microstructure and properties, and behave like ceramic materials manufactured by advanced ceramic processing approaches. This review aims to be a comprehensive description of the development of bioSiC ceramics: from wood templates and their microstructure to potential applications of bioSiC materials.
Agosto, 2017 · DOI: 10.1080/09506608.2017.1354429
Reactividad de Sólidos
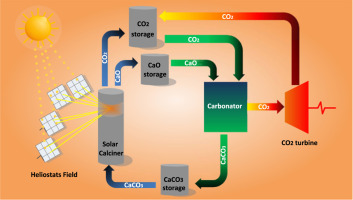
Large-scale high-temperature solar energy storage using natural minerals
Benitez-Guerrero, Monica; Sarrion, Beatriz; Perejon, Antonio; Sanchez-Jimenez, Pedro E.; Perez-Maqueda, Luis A.; Manuel Valverde, JoseSolar Energy Materials and Solar Cells, 168 (2017) 14-21
DOI: 10.1016/j.solmat.2017.04.013
Abstract
The present work is focused on thermochemical energy storage (TCES) in Concentrated Solar Power (CSP) plants by means of the Calcium-Looping (CaL) process using cheap, abundant and non-toxic natural carbonate minerals. CaL conditions for CSP storage involve calcination of CaCO3 in the solar receiver at relatively low temperature whereas carbonation of CaO is carried out at high temperature and high CO2 concentration to use the heat of reaction for power production by means of a CO2 closed power cycle. Under these conditions, large CaO particles derived from limestone to be used in industrial processes are rapidly deactivated due to pore plugging, which limits the extent of the reaction. This is favored by the relatively small pores of the CaO skeleton generated by low temperature calcination, the large thickness of the CaCO3 layer built upon the CaO surface and the very fast carbonation kinetics. On the other hand, at CaL conditions for CSP storage does not limit carbonation of CaO derived from dolomite (dolime). Dolime is shown to exhibit a high multicycle conversion regardless of particle size, which is explained by the presence of inert MgO grains that allow the reacting gas to percolate inside the porous particles.
Agosto, 2017 · DOI: 10.1016/j.solmat.2017.04.013
The epigraphic stela of Montoro (Cordoba): the earliest monumental script in Iberia?
Sanjuan, LG; Diaz-Guardamino, M; Wheatley, DW; Barra, JPV; Rodriguez, JAL; Rogerio-Candelera, MA; Erbez, AJ; Barker, D; Strutt, K; Ariza, MCAntiquity, 91 (2017) 916-932
DOI: 10.15184/aqy.2017.86
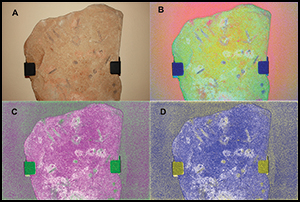
Abstract
A remarkable stela from Montoro, southern Spain, is unique in its morphology, epigraphic traits and landscape context. A programme of chemical characterisation, digital imaging, and geo-lithological and epigraphic analyses were conducted to determine its age and significance, and the results were integrated with data from archaeological investigations of the surrounding area. This multi-faceted approach allowed the stela to be interpreted within the context of early interactions between literate Mediterranean societies of the Late Bronze Age and Iron Age and non-literate Iberian societies. A key outcome of this research is a wider understanding of the complex patterns in the use and perception of early scripts.
Agosto, 2017 · DOI: 10.15184/aqy.2017.86
Nanotecnología en Superficies y Plasma
One-reactor plasma assisted fabrication of ZnO@TiO2 multishell nanotubes: assessing the impact of a full coverage on the photovoltaic performance
Filippin, Alejandro Nicolas; Macias-Montero, Manuel; Saghi, Zineb; Idigoras, Jesus; Burdet, Pierre; Sanchez-Valencia, Juan R.; Barranco, Angel; Migdley, Paul A.; Anta, Juan A.; Borras, AnaScientific Reports, 7 (2017) art 9621
DOI: 10.1038/s41598-017-09601-7
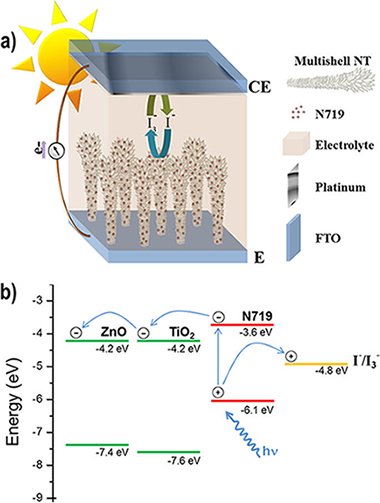
Abstract
This paper addresses the fabrication of vertically aligned ZnO@TiO2multishell nanotubes by a combined full vacuum-plasma approach at mild temperatures. The growth is carried out within the premises of a one-reactor approach, i.e. minimizing the number of vacuum chambers and sample transferences. In this way, the interface between ZnO and TiO2 is fully preserved from humidity thus increasing ZnO durability and stability. These nanostructures are studied by scanning electron microscopy (SEM), scanning transmission electron microscopy (STEM) and energy dispersive X-ray spectroscopy in STEM (EDX-STEM). High density one-dimensional arrays of these nanotubes formed on FTO substrates are applied as photoanode in a dye-sensitized solar cell (DSC). The evolution of the dye adsorption capacity and solar cells parameters are explored as a function of the crystallinity and thickness of the TiO2 shell. The results show the critical effect of a full coverage by TiO2 of ZnO core to explain the mixed results found in the literature.
Agosto, 2017 · DOI: 10.1038/s41598-017-09601-7
Reactividad de Sólidos
Flash sintering of highly insulating nanostructured phase-pure BiFeO3
Perez-Maqueda, LA; Gil-Gonzalez, E; Perejon, A; Lebrun, JM; Sanchez-Jimenez, PE; Raj, RJournal of the American Ceramic Society, 100 (2017) 3365-3369
DOI: 10.1111/jace.14990
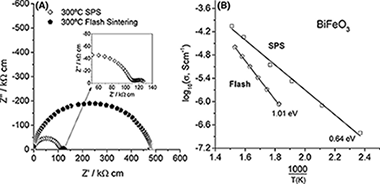
Abstract
We show that BiFeO3, that is electrically homogeneous, is a good insulator, and has a low dielectric constant (the properties desired in its applications), can be produced by flash sintering, which is nominally difficult to achieve by conventional and spark plasma sintering processes. The flash-sintered specimens had a uniform microstructure with a nanometric grain size of similar to 20 nm.
Agosto, 2017 · DOI: 10.1111/jace.14990
Materiales Avanzados
Thermal study of residues from greenhouse crops plant biomass
Morales, Laura; Garzon, Eduardo; Maria Martinez-Blanes, Jose; Jose Sanchez-Soto, PedroJournal of Thermal Analysis and Calorimetry, 129 (2017) 1111-1120
DOI: 10.1007/s10973-017-6243-2

Abstract
The principal aim of this work is to examine the effect of thermal treatments using a muffle furnace (static heating) and by simultaneous TG/DTA (dynamic heating) on selected greenhouse crops plant biomass investigated here as the first time. The effect of fractionation by sieving (<25 and <2.5 mm), preheating at 150 °C for 48 h and leaching with water on the thermal behavior has been studied. The observation of similar profiles of mass variation corresponding to several samples heated in air up to 1150 °C allows to conclude that particle size did not influence the thermal evolution, but the effect of heating cycle is evidenced. Thermal analysis in air of a representative sample showed the several mass variation steps and DTA exothermic effects produced by the complex thermal decomposition and pyrolysis of the organic matter. Elemental analysis (CHNS and O) of the starting samples and thermally treated revealed the effect of the temperature, with formation of ashes with lower C content from 44.37 to 0.70 mass% as a minimum after elimination of organic matter by heating. Leaching increased the thermal mass variation as an effect of elimination of water-soluble components. According to the present results, the size fractionation of the greenhouse crops biomass did not influence the results of elemental composition. The present study has provided results of interest concerning this biomass source of renewable energy originated by the remains of tomato (Solanum lycopersicum L.), being estimated the highest of all the biomass produced by the greenhouse crops agricultural industry in Almería (SE Spain).
Agosto, 2017 · DOI: 10.1007/s10973-017-6243-2
Materiales Ópticos Multifuncionales - Materiales Coloidales
Photonic Tuning of the Emission Color of Nanophosphor Films Processed at High Temperature
Geng, Dongling; Lozano, Gabriel; Calvo, Mauricio E.; Nunez, Nuria O.; Becerro, Ana I.; Ocana, Manuel; Miguez, HernanAdvanced Optical Materials, 5 (2017) art. 1700099
DOI: 10.1002/adom.201700099
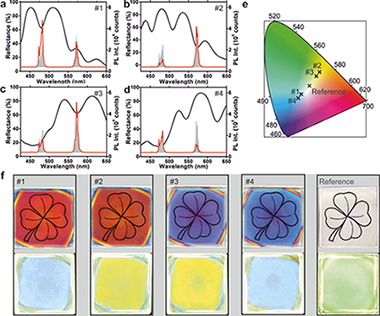
Abstract
Photonics offers new possibilities to tailor the photoluminescence process in phosphor-converted light emitting diodes. Herein, it is demonstrated that the emission color of thin layers of rare-earth doped nanocrystals can be strongly modulated in tunable spectral ranges using optical resonators specifically designed to this end. GdVO4:Dy3+ nanoparticles of controlled size and shape are synthesized using a solvothermal method with which highly transparent nanophosphor thin films are prepared. This paper designs and fabricates optical multilayers, which are transparent in the UV and resonant at the frequencies where the Dy3+ ions emit, to prove that the color coordinates of this emitter can be tuned from green to blue or yellow with unprecedented precision. Key to the achievement herein reported is the careful analysis of the structural and optical properties of thin nanophosphor layers with the processing temperature in order to achieve efficient photoluminescence while preserving the transparency of the film. The results open a new path for fundamental and applied research in solid-state lighting in which photonic nanostructures allow controlling the emission properties of state-of-the-art materials without altering their structure or chemical composition.
Julio, 2017 · DOI: 10.1002/adom.201700099
Nanotecnología en Superficies y Plasma
Low-Temperature Plasma Processing of Platinum Porphyrins for the Development of Metal Nanostructured Layers
Filippin, AN; Sanchez-Valencia, JR; Idigoras, J; Macias-Montero, M; Alcaire, M; Aparicio, FJ; Espinos, JP; Lopez-Santos, C; Frutos, F; Barranco, A; Anta, JA; Borras, AAdvanced Materials Interfaces, 4 (2017) 1601233
DOI: 10.1002/admi.201601233

Abstract
This article establishes the bases for a vacuum and plasma supported methodology for the fabrication at mild temperatures of nanostructured platinum in the form of porous layers and nanocolumns using platinum octaethylporphyrin as precursor. In addition, the application of these materials as tunable optical filters and nano-counterelectrodes is proved. On one hand, the transparency in the ultraviolet-visible-near infrared range can be adjusted precisely between 70% and 1% by tuning the deposition and processing conditions, obtaining a high spectral planarity. Deviations of the spectra from an ideal flat filter are below 4%, paving the way to the fabrication of neutral density filters. The transparency limit values yield a sheet resistivity of approximate to 1350 and 120 Omega square(-1), respectively. On the other hand, the catalytic properties of the nanostructures are further demonstrated by their implementation as counterelectrodes of excitonic solar cells surpassing the performance of commercial platinum as counterelectrode in a 20% of the overall cell efficiency due to simultaneous enhancement of short-circuit photocurrent and open-circuit photovoltage. One of the most interesting features of the developed methodology is its straightforward application to other metal porphyrins and phthalocyanines readily sublimable under mild vacuum and temperature conditions.
Julio, 2017 · DOI: 10.1002/admi.201601233
Reactividad de Sólidos
Characterization of of mechanosynthesized Bi1-xSmxFeO3 samples unencumbered by secondary phases or compositional inhomogeneity
Gil-Gonzalez, E; Perejon, A; Sanchez-Jimenez, PE; Hayward, MA; Criado, JM; Sayagues, MJ; Perez-Maqueda, LAJournal of Alloys and Compounds, 711 (2017) 541-551
DOI: 10.1016/j.jallcom.2017.03.289
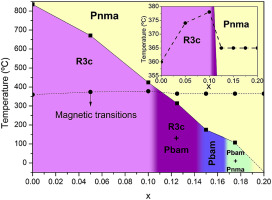
Abstract
High-quality samples, in terms of phase purity and dielectric properties, of composition Bi1-xSmxFeO3 (0.05 <= x <= 0.20) have for the first time been prepared by mechanosynthesis. Close inspection of the powder diffraction data, analysis via Rietveld refinement and TEM microscopy demonstrates that the Bi1-xSmxFeO3 samples contain only perovskite phases. Additionally, by a combination of Rietveld analysis, TEM, DSC, temperature-dependent XRD and permittivity data a tentative phase diagram has been proposed where the high temperature paraelectric phase Pnma has been confirmed for samarium substituted BiFeO3. Regarding the physical properties, the samples resulted to be electrically homogenous and highly insulating at room temperature, suggesting that other sources of conductivity, such as mixed valence of Fe associated with possible oxygen non-stoichiometry, have been avoided during the samples synthesis. In spite of the high quality of the samples, the dielectric and magnetic behaviour of the Bi1-xSmxFeO3 samples change only modestly on Sm substitution, with neither a great change in the resistivity or remnant magnetisation of Sm substituted samples in comparison with BiFeO3.
Julio, 2017 · DOI: 10.1016/j.jallcom.2017.03.289
Nanotecnología en Superficies y Plasma
Formation of Subsurface W5+ Species in Gasochromic Pt/WO3 Thin Films Exposed to Hydrogen
Castillero, Pedro; Rico-Gavira, Victor; Lopez-Santos, Carmen; Barranco, Angel; Perez-Dieste, Virginia; Escudero, Carlos; Espinos, Juan P.; Gonzalez-Elipe, Agustin R.Journal of Physical Chemistry C, 121 (2017) 15719-15727
DOI: 10.1021/acs.jpcc.7b03385
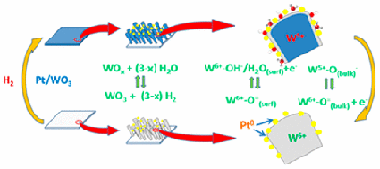
Abstract
M/WO3 (M = Pt, Pd) systems formed by a porous WO3 thin film decorated by metal nanoparticles are known for their reversible coloring upon exposure to H2 at room temperature. In this work, this gasochromic behavior is investigated in situ by means of near-ambient photoemission (NAPP). Pt/WO3 systems formed by very small Pt nanoparticles (10 ± 1 nm average size) incorporated in the pores of nanocolumnar WO3 thin films prepared by magnetron sputtering at an oblique angle have been exposed to a small pressure of hydrogen at ambient temperature. The recorded UV–vis transmission spectra showed the reversible appearance of a very intense absorption band responsible for the blue coloration of these gasochromic films. In an equivalent experiment carried out in the NAPP spectrometer, W 4f, O 1s, Pt 4f, and valence band photoemission spectra have been recorded at various photon energies to follow the evolution of the reduced tungsten species and hydroxyl groups formed upon film exposure to hydrogen. The obtained results are compared with those of a conventional X-ray photoemission study after hydrogen exposure between 298 and 573 K. As investigated by NAPP, the gasochromic behavior at 298 K is accounted for by a reaction scheme in which hydrogen atoms resulting from the dissociation of H2 onto the Pt nanoparticles are spilt over to the WO3 substrate where they form surface OH–/H2O species and subsurface W5+ cations preferentially located in buried layers of the oxide network.
Julio, 2017 · DOI: 10.1021/acs.jpcc.7b03385
Materiales y Procesos Catalíticos de Interés Ambiental y Energético
Cobalt Carbide Identified as Catalytic Site for the Dehydrogenation of Ethanol to Acetaldehyde
A. Rodríguez-Gómez; J.P. Holgado; A. CaballeroACS Catalysis, 7 (2017) 5243-5247
DOI: 10.1021/acscatal.7b01348
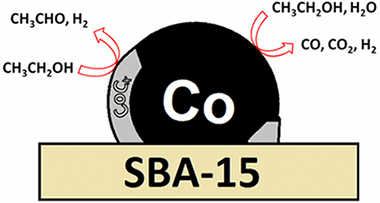
Abstract
Two cobalt catalysts, Co/SBA-15 and Co/SiO2, have been studied in steam reforming of ethanol (SRE). Besides the steam reforming products, ethoxide dehydrogenation to acetaldehyde is observed as one of the main reactions. Although by hydrogen treatment cobalt is reduced to the metallic state, under SRE conditions, a phase appears that has been identified as cobalt carbide and correlates with acetaldehyde production. These findings provide insights about the catalytic sites, for SRE, in cobalt catalysts. Comparison with previous results shows that these conclusions are not translatable to other cobalt catalysts, stressing the importance of the support on the catalytic behavior of cobalt.
Julio, 2017 · DOI: 10.1021/acscatal.7b01348
Materiales de Diseño para la Energía y Medioambiente
Cs+ immobilization by designed micaceous adsorbent under subcritical conditions
Osuna, FJ; Cota, A; Pavon, E; Pazos, MC; Alba, MDApplied Clay Science, 143 (2017) 293-299
DOI: 10.1016/j.clay.2017.03.041
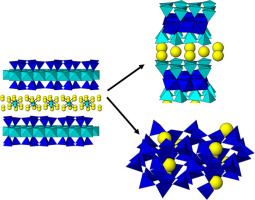
Abstract
The adsorption of Cs+ by clay minerals is a complicate process, being cation exchange and frayed-edge sites the major mechanisms that govern it. However, environmental variables have a significant impact on the process. In this work, the influence of the temperature and time in the cesium adsorption capacity of Na-Mica-n (n = 2 and 4) have been explored under subcritical conditions. Those synthetic micas were able to immobilize cations Cs+ combining adsorption at nonspecific sites, at specific sites and chemical reaction. The distribution constant of Cs+ was larger in the Na-Mica-2 denoting a higher concentration of specific adsorption sites when layer charge decreased.
Julio, 2017 · DOI: 10.1016/j.clay.2017.03.041
Nanotecnología en Superficies y Plasma - Tribología y Protección de Superficies
Plasma assisted deposition of single and multistacked TiO2 hierarchical nanotube photoanodes
Filippin, AN; Sanchez-Valencia, JR; Idigoras, J; Rojas, TC; Barranco, A; Anta, JA; Borras, ANanoscale, 9 (2017) 8133-8141
DOI: 10.1039/c7nr00923b
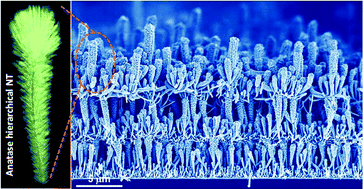
Abstract
We present herein an evolved methodology for the growth of nanocrystalline hierarchical nanotubes combining physical vapor deposition of organic nanowires (ONWs) and plasma enhanced chemical vacuum deposition of anatase TiO2 layers. The ONWs act as vacuum removable 1D and 3D templates, with the whole process occurring at temperatures ranging from RT to 250 degrees C. As a result, a high density of hierarchical nanotubes with tunable diameter, length and tailored wall microstructures are formed on a variety of processable substrates as metal and metal oxide films or nanoparticles including transparent conductive oxides. The reiteration of the process leads to the development of an unprecedented 3D nanoarchitecture formed by stacking the layers of hierarchical TiO2 nanotubes. As a proof of concept, we present the superior performance of the 3D nanoarchitecture as a photoanode within an excitonic solar cell with efficiencies as high as 4.69% for a nominal thickness of the anatase layer below 2.75 mu m. Mechanical stability and straightforward implementation in devices are demonstrated at the same time. The process is extendable to other functional oxides fabricated by plasma-assisted methods with readily available applications in energy harvesting and storage, catalysis and nanosensing.
Julio, 2017 · DOI: 10.1039/c7nr00923b
Nanotecnología en Superficies y Plasma
Surface chemistry and germination improvement of Quinoa seeds subjected to plasma activation
Gomez-Ramierez, A.; Lopez-Santos, C.; Cantos, M.; Garcia, J. L.; Molina, R.; Cotrino, J.; Espinos, J. P.; Gonzalez-Elipe, A. R.Scientific Reports, 7 (2017) art. 5924
DOI: 10.1038/s41598-017-06164-5

Abstract
Plasma treatment is recognized as a suitable technology to improve germination efficiency of numerous seeds. In this work Quinoa seeds have been subjected to air plasma treatments both at atmospheric and low pressure and improvements found in germination rate and percentage of success. Seed water uptake by exposure to water vapor, although slightly greater for plasma treated seeds, did not justify the observed germination improvement. To identify other possible factors contributing to germination, the chemical changes experienced by outer parts of the seed upon plasma exposure have been investigated by X-ray photoemission spectroscopy (XPS) and scanning electron microscopy (SEM-EDX). XPS revealed that the outer layers of the Quinoa plasma treated seeds were highly oxidized and appeared enriched in potassium ions and adsorbed nitrate species. Simultaneously, SEM-EDX showed that the enrichment in potassium and other mineral elements extended to the seed pericarp and closer zones. The disappearance from the surface of both potassium ions and nitrate species upon exposure of the plasma treated seeds to water vapor is proposed as a factor favoring germination. The use of XPS to study chemical changes at seed surfaces induced by plasma treatments is deemed very important to unravel the mechanisms contributing to germination improvement.
Julio, 2017 · DOI: 10.1038/s41598-017-06164-5
Reactividad de Sólidos
Non-isothermal Characterization of the Precipitation Hardening of a Cu-11Ni-19Zn-1Sn Alloy
Donoso, E; Dianez, MJ; Criado, JM; Espinoza, R; Mosquera, EMetallurgical and Materials Transactions A-Physical Metallurgy and Materials Science, 48A (2017) 3090-3095
DOI: 10.1007/s11661-017-4063-4

Abstract
The precipitation hardening of a Cu-11Ni-19Zn-1Sn alloy has been studied by means of Differential Scanning Calorimetry (DSC), High-Resolution Transmission Electron Microscopy (HRTEM), and hardness measurements. The calorimetric curves, in the range of temperatures analyzed, show the presence of one exothermic reaction followed by an endothermic one. The exothermic DSC peak is due to the segregation of Cu2NiZn precipitates and it is associated to a noticeable improvement of the mechanical properties of the alloy. The endothermic effect is associated to the dissolution of the Cu2NiZn precipitates into the copper matrix for restoring the starting Cu-11Ni-19Zn-1Sn homogeneous solid solution. The reaction mechanisms of these processes have been proposed from the kinetic analysis of the exothermic and endothermic DSC signals. The results obtained point out that tin plays a decisive role on the precipitation hardening of the alloy, because age hardening is not observed in the case of a Cu-Ni-Zn ternary alloy of similar composition.
Junio, 2017 · DOI: 10.1007/s11661-017-4063-4
Nanotecnología en Superficies y Plasma
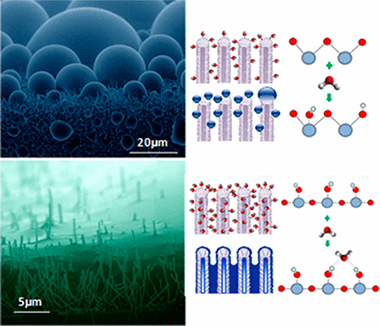
In Situ Determination of the Water Condensation Mechanisms on Superhydrophobic and Superhydrophilic Titanium Dioxide Nanotubes
Macias-Montero, Manuel; Lopez-Santos, Carmen; Nicolas Filippin, A.; Rico, Victor J.; Espinos, Juan P.; Fraxedas, Jordi; Perez-Dieste, Virginia; Escudero, Carlos; Gonzalez-Elipe, Agustin R.; Borras, AnaLangmuir, 33 (2017) 6449-6456
DOI: 10.1021/acs.langmuir.7b00156
Abstract
One-dimensional (1D) nanostructured surfaces based on high-density arrays of nanowires and nanotubes of photoactive titanium dioxide (TiO2) present a tunable wetting behavior from superhydrophobic to superhydrophilic states. These situations are depicted in a reversible way by simply irradiating with ultraviolet light (superhydrophobic to superhydrophilic) and storage in dark. In this article, we combine in situ environmental scanning electron microscopy (ESEM) and near ambient pressure photoemission analysis (NAPP) to understand this transition. These experiments reveal complementary information at microscopic and atomic level reflecting the surface wettability and chemical state modifications experienced by these 1D surfaces upon irradiation. We pay special attention to the role of the water condensation mechanisms and try to elucidate the relationship between apparent water contact angles of sessile drops under ambient conditions at the macroscale with the formation of droplets by water condensation at low temperature and increasing humidity on the nanotubes surfaces. Thus, for the as-grown nanotubes, we reveal a metastable and superhydrophobic Cassie state for sessile drops that tunes toward water dropwise condensation at the microscale compatible with a partial hydrophobic Wenzel state. For the UV-irradiated surfaces, a filmwise wetting behavior is observed for both condensed water and sessile droplets. NAPP analyses show a hydroxyl accumulation on the as-grown nanotubes surfaces during the exposure to water condensation conditions, whereas the water filmwise condensation on a previously hydroxyl enriched surface is proved for the superhydrophilic counterpart.
Junio, 2017 · DOI: 10.1021/acs.langmuir.7b00156
Reactividad de Sólidos
The Oxy-CaL process: A novel CO2 capture system by integrating partial oxy-combustion with the Calcium-Looping process
Ortiz, C; Valverde, JM; Chacartegui, R; Benitez-Guerrero, M; Perejon, A; Romeo, LMApplied Energy, 196 (2017) 1-17
DOI: 10.1016/j.apenergy.2017.03.120
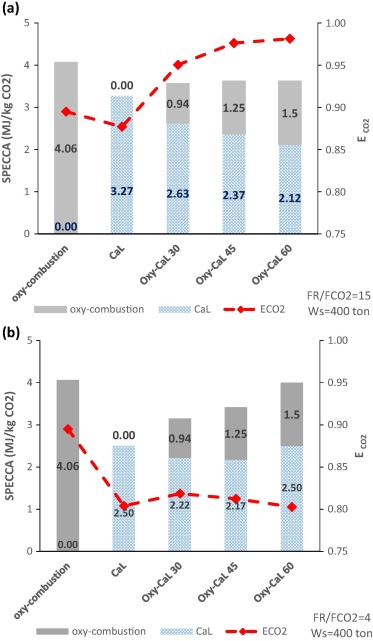
Abstract
This paper proposes a novel CO2 capture technology from the integration of partial oxy-combustion and the Calcium -Looping capture process based on the multicycle carbonation/calcination of limestone derived CaO. The concentration of CO2 in the carbonator reactor is increased by means of partial oxycombustion, which enhances the multicycle CaO conversion according to thermogravimetric analysis results carried out in our work, thus improving the CO2 capture efficiency. On the other hand, energy consumption for partial oxy-combustion is substantially reduced as compared to total oxy-combustion. All in all, process simulations indicate that the integration of both processes has potential advantages mainly regarding power plant flexibility whereas the overall energy penalty is not increased. Thus, the resulting energy consumption per kilogram of CO2 avoided is kept smaller than 4 MI/kg CO2, which remains below the typical values reported for total oxy-combustion and amine based CO2 capture systems whereas CO2 capture efficiency is enhanced in comparison with the Calcium -Looping process.
Junio, 2017 · DOI: 10.1016/j.apenergy.2017.03.120
Materiales de Diseño para la Energía y Medioambiente
Failure mode and effect analysis of a large scale thin-film CIGS photovoltaic module
Delgado-Sanchez, JM; Sanchez-Cortezon, E; Lopez-Lopez, C; Aninat, R; Alba, MDEngireering failure analysis, 76 (2017) 55-60
DOI: 10.1016/j.engfailanal.2017.02.004
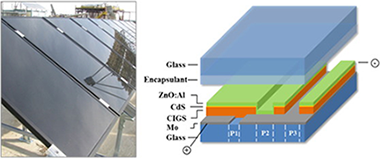
Abstract
The efficiency of thin-film CIGS based cells at the laboratory scale is now getting closer to conventional Silicon technologies. As a consequence, the long-term stability of CIGS is now one of the main challenges left to address in order to assess its potential as an alternative for photovoltaic plants. This paper reports an overview of the critical risks for the commercial viability of the CIGS thin-film technology. The key causes of the potential failures of this technology are determined through the Failure Mode Analysis and Effects (FMEA) methodology. To validate the results obtained from the FMEA, aging tests and outdoor monitoring were also carried out. Based on the results obtained, we argue that the encapsulation material is the main cause of degradation in CIGS modules.
Junio, 2017 · DOI: 10.1016/j.engfailanal.2017.02.004
Reactividad de Sólidos
A novel, simple and rapid route to the synthesis of boron cabonitride nanosheets: combustive gaseous unfolding
Jalaly, Maisam; Jose Gotor, Francisco; Semnan, Masih; Jesus Sayagues, MariaScientific Reports, 7 (2017) art. 3453
DOI: 10.1038/s41598-017-03794-7
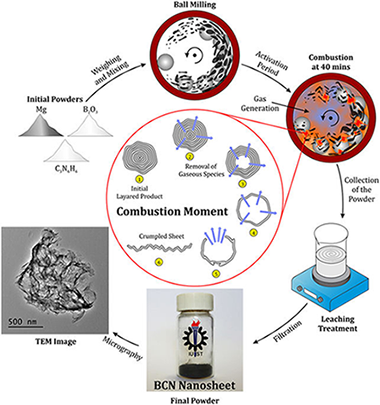
Abstract
The ternary compound boron carbonitride (BCN) was synthesized in the form of few-layer nanosheets through a mechanically induced self-sustaining reaction (MSR). Magnesium was used to reduce boron trioxide in the presence of melamine in a combustive manner. The process to form the nanostructured material was very rapid (less than 40 min). The prepared powder was investigated by various techniques such as X-ray diffraction (XRD), Fourier Transform infrared (FTIR), Micro-Raman spectroscopy, X-ray photoelectron spectroscopy (XPS), high-resolution transmission electron microscopy (HRTEM), and electron energy loss spectroscopy (EELS). The thermal stability and the optical behavior of the BCN nanosheets were also studied by thermal analysis and UV-vis spectroscopy, respectively. The formation mechanism of the nanosheet morphology was described in detail.
Junio, 2017 · DOI: 10.1038/s41598-017-03794-7
Nanotecnología en Superficies y Plasma
A compact and portable optofluidic device for detection of liquid properties and label-free sensing
Lahoz, F; Martin, IR; Walo, D; Gil-Rostra, J; Yubero, F; Gonzalez-Elipe, ARJournal of Physics D: Applied Physics, 50 (2017) 21
DOI: 10.1088/1361-6463/aa6cdd
Abstract
Optofluidic lasers have been widely investigated over the last few years mainly because they can be easily integrated in sensor devices. However, high power pulse lasers arc required as excitation sources, which, in practice, limit the portability of the system. Trying to overcome some of these limitations, in this paper we propose the combined use of a small CW laser with a Fabry-Perot optofluidic planar microcavity showing high sensitivity and versatility for detection of liquid properties and label-free sensing. Firstly, a fluorescein solution in ethanol is used to demonstrate the high performances of the FP microcavity as a temperature sensor both in the laser (high pump power above laser threshold) and in the fluorescence (low pump power) regimes. A shift in the wavelength of the resonant cavity modes is used to detect changes in the temperature and our results show that high sensitivities could be already obtained using cheap and portable CW diode lasers. In the second part of the paper, the demonstration of this portable device for label-free sensing is illustrated under low CW pumping. The wavelength positions of the optolluidic resonant modes are used to detect glucose concentrations in water solutions using a protein labelled with a fluorescent dye as the active medium.
Junio, 2017 · DOI: 10.1088/1361-6463/aa6cdd
Materiales y Procesos Catalíticos de Interés Ambiental y Energético
Analysis of Ni species formed on zeolites, mesoporous silica and alumina supports and their catalytic behavior in the dry reforming of methane
Drobna, Helena; Kout, Martin; Soltysek, Agnieszka; Gonzalez-Delacruz, Victor M.; Caballero, Alfonso; Capek, LiborReaction Kinetics Mechanisms and Catalysis, 121 (2017) 255-274
DOI: 10.1007/s11144-017-1149-3

Abstract
The presented investigation is focused on the analysis of Ni species formed on microporous (zeolites MFI and FAU) and mesoporous materials (Al-MCM- 41 and SBA-15) and alumina supports and their catalytic behavior in the dry reforming of methane. The paper lays emphasis on the relationship between the catalytic behavior of Ni-based catalysts and their textural/structural properties. Ni-based catalysts were prepared by wet impregnation (11 wt% of Ni) followed by calcination in air and reduction in hydrogen. The properties of Ni-based catalysts were also compared prior and after the catalytic tests. The critical role was played by the high value of the specific surface area and the high strength of the interaction between the Ni species and the support, which both determined the high dispersion and stability of metal Ni-0 particles. Ni-Al-MCM-41 and Ni-SBA-15 showed the values of the conversion of CO2 and CH4 above 90% (stable during 12 h). Slightly lower values of the conversion of CO2 and CH4 were observed over Ni-Al2O3 (also stable during 12 h). In contrast to these materials, Ni-MFI and Ni-FAU exhibited the worse metallic Ni-0 particles dispersion and very bad catalytic behavior.
Junio, 2017 · DOI: 10.1007/s11144-017-1149-3
Nanotecnología en Superficies y Plasma
Impact of moisture on efficiency-determining electronic processes in perovskite solar cells
Salado, Manuel; Contreras-Bernal, Lidia; Calio, Laura; Todinova, Anna; Lopez-Santos, Carmen; Ahmad, Shahzada; Borras, Ana; Idigoras, Jesus; Anta, Juan A.Journal of Materials Chemistry A, 5 (2017) 10917-10927
DOI: 10.1039/c7ta02264f
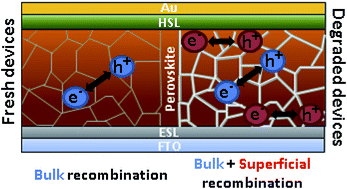
Abstract
Moisture-induced degradation in perovskite solar cells was thoroughly investigated by structural (SEM, EDS, XRD and XPS) and device characterization (impedance and intensity modulated photocurrent spectroscopy) techniques. Both the influence of the perovskite composition and the nature of the hole selective material were analyzed. The degradation rate was found to be significantly slower for mixed perovskites and P3HT-based devices. However, for a fixed degradation degree (defined as a 50% drop from the initial photocurrent), all configurations show similar features in small-perturbation analysis. Thus, a new mid-frequency signal appears in the impedance response, which seems to be related to charge accumulation at the interfaces. In addition, faster recombination, with a more important surface contribution, and slower transport were clearly inferred from our results. Both features can be associated with the deterioration of the contacts and the formation of a higher number of grain boundaries.
Junio, 2017 · DOI: 10.1039/c7ta02264f
Materiales para Bioingeniería y Regeneración Tisular
High surface area biopolymeric-ceramic scaffolds for hard tissue engineering
Romero-Sanchez, LB; Borrego-Gonzalez, S; Diaz-Cuenca, ABiomedical Physics & Engineering Express, 3 (2017) art UNSP 035012
DOI: 10.1088/2057-1976/aa7001
Abstract
The development of scaffolds mimicking native bone tissue composition and structure is a challenge in bone tissue engineering. 3D scaffolds with both an interconnected macropore structure and nanotextured surfaces are required. However, 3D scaffolds processed by microfabrication usually lack of nanotextured surface, while nanotextured materials generated by bottom-up nanofabrication are difficult to process conforming scaffolds having well interconnected microsized cavities. In this work, the processing of reticulated (macropore interconnected) structures using nanostructured precursors has been performed to improve the mechanical properties of the scaffolds. The application of a fibrillar collagen coating, using less than 1 wt% collagen per scaffold, has allow a significant increase of the compressive strength while preserving a high surface area and nanopore accessibility. Besides, the fibrillar nanostructured collagen coating promotes hydroxyapatite mineralization. Two different collagen-coating procedures are applied showing interesting differences in terms of mechanical performance.
Junio, 2017 · DOI: 10.1088/2057-1976/aa7001
Nanotecnología en Superficies y Plasma
About the enhancement of chemical yield during the atmospheric plasma synthesis of ammonia in a ferroelectric packed bed reactor
Gomez-Ramirez, Ana; Montoro-Damas, Antonio M.; Cotrino, Jose; Lambert, Richard M.; Gonzalez-Elipe, Agustin R.Plasma Processes and Polymers, 14 (2017) e1600081
DOI: 10.1002/ppap.201600081

Abstract
Plasma reactions offer an attractive alternative route for the synthesis of a variety of valuable chemical compounds. Here we investigate the parameters that determine the efficiency of ammonia synthesis in a ferroelectric packed bed dielectric barrier discharge (DBD) reactor. The effects of varying the operating frequency, the size of the ferroelectric pellets and the inter-electrode distance have been systematically studied. Under optimised conditions nitrogen conversions in excess of 7% were achieved, higher than those previously obtained using DBD reactors. These findings are discussed with respect to variations in the electrical characteristics of the reactor under operating conditions and in the light of emission spectra obtained as a function of reactant flow rates. These encouraging results signpost future developments that could very substantially improve the efficiency of ammonia synthesis by means of DBD technology.
Junio, 2017 · DOI: 10.1002/ppap.201600081
Reactividad de Sólidos
Effect of the impact energy on the chemical homogeneity of a (Ti,Ta,Nb)(C,N) solid solution obtained via a mechanically induced self-sustaining reaction
de La Obra, AG; Gotor, FJ; Chicardi, EJournal of Alloys and Compounds, 708 (2017) 1008-1017
DOI: 10.1016/j.jallcom.2017.03.109
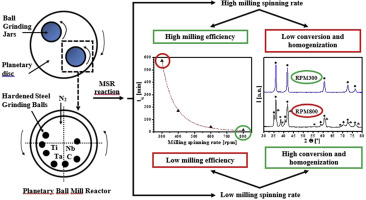
Abstract
A titanium-tantalum-niobium carbonitride solid solution, (Ti,Ta,Nb)(C,N), was synthesised in a planetary mill via a mechanochemical process that involves a mechanically induced self-sustaining reaction (MSR) from stoichiometric Ti/Ta/Nb/C mixtures that are milled under a nitrogen atmosphere. The influence of the spinning rate of the planetary mill, which determines the impact energy of the milling process, on the ignition time (t(ig)) of the MSR process as well as the chemical homogeneity of the final product was analysed. The results indicated that the dependence of tig on the spinning rate followed a potential function with a potential factor of 4.85, implying a remarkable reduction in the milling time required to induce the self-sustaining reaction at increasing spinning rates (i.e., from 4200 min at 200 rpm to 15 min at 800 rpm). However, the chemical and structural characterisation of the obtained products at ignition without any extra milling treatment indicated that a single solid solution phase was only obtained at the lowest spinning rates (i.e., less than 300 rpm). At increasing rates, the relative amount of the intended solid solution phase continuously decreased, and new undesirable secondary phases were formed. Despite the long milling times required for the milling experiments that were performed at the slowest spinning rates, iron contamination from the milling media was negligible due to the low intensity milling regime.
Junio, 2017 · DOI: 10.1016/j.jallcom.2017.03.109
Propiedades mecánicas, modelización y caracterización de cerámicos avanzados
High-temperature creep of carbon nanofiber-reinforced and graphene oxide-reinforced alumina composites sintered by spark plasma sintering
Cano-Crespo, Rafael; Malmal Moshtaghioun, Bibi; Gomez-Garcia, Diego; Dominguez-Rodriguez, Arturo; Moreno, RodrigoCeramics International, 43 (2017) 7136-7141
DOI: 10.1016/j.ceramint.2017.02.146

Abstract
Alumina (Al2O3) ceramic composites reinforced with either graphene oxide (GO) or carbon nanofibers (CNFs) were prepared using Spark Plasma Sintering. The effects of GO and CNFs on the microstructure and in consequence on their mechanical properties were investigated. The microstructure of the sintered materials have been characterized quantitatively prior to and after the creep experiments in order to discover the deformation mechanism. Graphene-oxide reinforced alumina composites were found to be more creep resistant than carbon nanofibers-reinforced alumina ones or monolithic alumina with the same grain size distribution. In all the cases, grain boundary sliding was identified as the deformation mechanism
Junio, 2017 · DOI: 10.1016/j.ceramint.2017.02.146
Nanotecnología en Superficies y Plasma
Energy-Sensitive Ion- and Cathode-Luminescent Radiation-Beam Monitors Based on Multilayer Thin-Film Designs
Gil-Rostra, Jorge; Ferrer, Francisco J.; Pedro Espinos, Juan; Gonzalez-Elipe, Agustin R.; Yubero, FranciscoACS Applied Materials & Interfaces, 9 (2017) 16313-16320
DOI: 10.1021/acsami.7b01175
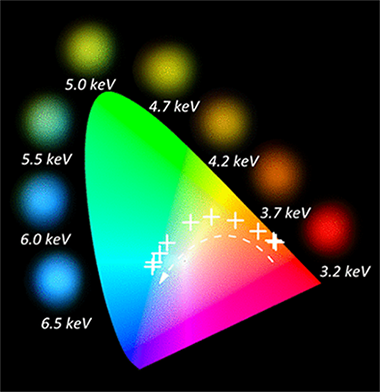
Abstract
A multilayer luminescent design concept is presented to develop energy sensitive radiation-beam monitors on the basis of colorimetric analysis. Each luminescent layer within the stack consists of rare-earth-doped transparent oxides of optical quality and a characteristic luminescent emission under excitation with electron or ion beams. For a given type of particle beam (electron, protons, alpha particles, etc.), its penetration depth and therefore its energy loss at a particular buried layer within the multilayer stack depend on the energy of the initial beam. The intensity of the luminescent response of each layer is proportional to the energy deposited by the radiation beam within the layer, so characteristic color emission will be achieved if different phosphors are considered in the layers of the luminescent stack. Phosphor doping, emission efficiency, layer thickness, and multilayer structure design are key parameters relevant to achieving a broad colorimetric response. Two case examples are designed and fabricated to illustrate the capabilities of these new types of detector to evaluate the kinetic energy of either electron beams of a few kilo-electron volts or a particles of alpha few mega-electron volts.
Mayo, 2017 · DOI: 10.1021/acsami.7b01175
Fotocatálisis Heterogénea: Aplicaciones
NO photooxidation with TiO2 photocatalysts modified with gold and platinum
Rodriguez, MJH; Melian, EP; Santiago, DG; Diaz, OG; Navio, JA; Rodriguez, JMDApplied Catalysis B-Environmental, 205 (2017) 148-157
DOI: 10.10161/j.apcatb.2016.12.006
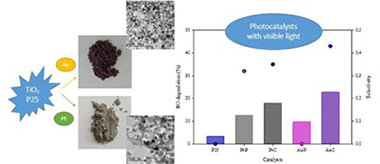
Abstract
In this study, a comparative analysis is made of TiO2 modified with Pt or Au in NO photoxidation under different radiation and humidity conditions. The metals were deposited on the TiO2 surface using two methods, photodeposition and chemical reduction. All catalysts were supported on borosilicate 3.3 plates using a dip-coating technique. These modified photocatalysts were characterized by X-ray diffraction analysis (XRD), UV-vis diffuse reflectance spectra (DRS), Brunauer-Emmett-Teller measurements (BET), transmission electron microscopy (TEM) and X-ray photoelectron spectrum analysis (XPS). It was found from the XPS results that Pt and oxidized Pt species coexist on the samples obtained by photodeposition and chemical reduction. In the case of Au, though other oxidation states were also detected the dominant oxidation state for both catalysts is Au. TEM results showed most Au-C particles are below 5 nm, whereas for Au-P the nanoparticles are slightly bigger. With UV irradiation, the Pt modified catalysts do not show any significant improvement in NO photocatalytic oxidation in comparison with the unmodified P25. For Au, both modified photocatalysts (Au-P and Au-C) exceed the photocatalytic efficiency of the unmodified P25, with Au-C giving slightly better results. The incorporation of metals on the TiO2 increases its activity in the visible region.
Mayo, 2017 · DOI: 10.10161/j.apcatb.2016.12.006
Reactividad de Sólidos
New findings on thermal degradation properties of fluoropolymers
Liu, SE; Zhou, WL; Yan, QL; Qi, XF; An, T; Perez-Maqueda, LA; Zhao, FQJournal of Thermal Analysis and Calorimetry, 128 (2017) 675-685
DOI: 10.1007/s10973-016-5963-z
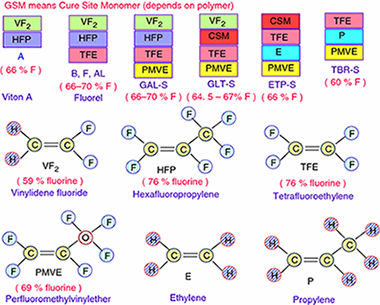
Abstract
In this paper, the thermal degradation properties of Viton A and Fluorel are investigated by both isoconversional and combined kinetic analysis methods using non-isothermal thermogravimetry technique. It has been found that the heating rate has little affect on the degradation residue of Fluorel and Viton A, where around 1.3% char was formed for Fluorel and 3.5% for Viton A. Different from the literature, the decomposition of Viton A should be considered as an overlapped dehydrofluorination and carbon chain scission process, with activation energy of 214 +/- 11 and 268 +/- 13 kJ mol(-1), respectively. The effect of dehydrofluorination on degradation of Fluorel is not so significant due to low content of H, and hence, it could be considered as a single-step mechanism with average activation energy of 264 +/- 14 kJ mol(-1). The thermal stability of Fluorel is much better than that of Viton A, and the predicted half-life is around 218 min for Fluorel and 49 min for Viton A at 420 A degrees C, which are consistent with experimental values. If using a single-step model as in the literature for Viton A, its half-life at 420 A degrees C would be underestimated for > 20%.
Mayo, 2017 · DOI: 10.1007/s10973-016-5963-z
Nanotecnología en Superficies y Plasma
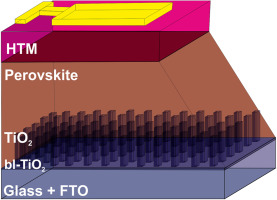
1-dimensional TiO2 nano-forests as photoanodes for efficient and stable perovskite solar cells fabrication
Salado, M; Oliva-Ramirez, M; Kazim, S; Gonzalez-Elipe, AR; Ahmad, SNano Energy, 35 (2017) 215-222
DOI: 10.1016/j.nanoen.2017.03.034
Abstract
During the last years, perovskite solar cells have gained increasing interest among the photovoltaic community, in particularly after reaching performances at par with mature thin film based PV. This rapid evolution has been fostered by the compositional engineering of perovskite and new device architectures. In the present work, we report the fabrication of perovskite solar cells based on highly ordered 1-dimensional vertically oriented TiO2 nano-forests. These vertically oriented porous TiO2 photoanodes were deposited by physical vapor deposition in an oblique angle configuration, a method which is scalable to fabricate large area devices. Mixed (MA0.15FA0.85)Pb(I0.85Br0.15)3 or triple cation Cs0.05(MA0.15FA0.85)0.95Pb(I0.85Br0.15)3 based perovskites were then infiltrated into these 1-dimensional nanostructures and power conversion efficiencies of 16.8% along with improved stability was obtained. The devices fabricated using 1D-TiO2 were found to be more stable compare to the classical 3-dimensional TiO2 photoanodes prepared by wet chemistry. These 1-D photoanodes will be of interest for scaling up the technology and in other opto-electrical devices as they can be easily fabricated utilizing industrially adapted methodologies.
Mayo, 2017 · DOI: 10.1016/j.nanoen.2017.03.034
Reactividad de Sólidos
Structure evolution in the LaMn1 − xFexO3 + δ system by Rietveld analysis
Cordoba, J. M.; Ponce, M.; Sayagues, M. J.Solis State Ionics, 303 (2017) 132-137
DOI: 10.1016/j.ssi.2017.02.020
Abstract
The synthesis of LaMn1 − xFexO3 + δ (0 ≤ x ≤ 1) solid solutions perovskite powder was carried out using high-energy milling from the constituent oxides, and further crystallization by high temperature treatment. The compositions of the crystalline phases as a function of x were determined by X-ray powder diffraction using a Rietveld refinement. The relationship between composition and structure was covered. This showed that LaMn1 − xFexO3 + δ exists with the rhombohedral structure (R-3c, 167) only below x = 0.3 and with the orthorhombic structure (Pnma, 62) over x = 0.7. The rhombohedral phase coexists with the orthorhombic phase between 0.4 < x < 0.6.
Mayo, 2017 · DOI: 10.1016/j.ssi.2017.02.020
Reactividad de Sólidos
CO2 capture performance of Ca-Mg acetates at realistic Calcium Looping conditions
Miranda-Pizarro, J; Perejon, A; Valverde, JM; Perez-Maqueda, LA; Sanchez-Jimenez, PEFuel, 196 (2017) 497-507
DOI: 10.1016/j.fuel.2017.01.119

Abstract
The Calcium Looping (CaL) process, based on the cyclic carbonation/calcination of CaO, has emerged in the last years as a potentially low cost technique for CO2 capture at reduced energy penalty. In the present work, natural limestone and dolomite have been pretreated with diluted acetic acid to obtain Ca and Ca-Mg mixed acetates, whose CO2 capture performance has been tested at CaL conditions that necessarily imply sorbent regeneration under high CO2 partial pressure. The CaL multicycle capture performance of these sorbents has been compared with that of CaO directly derived from limestone and dolomite calcination. Results show that acetic acid pretreatment of limestone does not lead to an improvement of its capture capacity, although it allows for a higher calcination efficiency to regenerate CaO at reduced temperatures (similar to 900 degrees C) as compared to natural limestone (>similar to 930 degrees C). On the other hand, if a recarbonation stage is introduced before calcination to reactivate the sorbent, a significantly higher residual capture capacity is obtained for the Ca -Mg mixed acetate derived from dolomite as compared to either natural dolomite or limestone. The main reason for this behavior is the enhancement of carbonation in the solid-state diffusion controlled phase. It is argued that the presence of inert MgO grains in the mixed acetate with reduced segregation notably promotes solid state diffusion of ions across the porous structure created after recarbonation.
Mayo, 2017 · DOI: 10.1016/j.fuel.2017.01.119
Nanotecnología en Superficies y Plasma
Non-enzymatic hydrogen peroxide detection at NiO nanoporous thin film-electrodes prepared by physical vapor deposition at oblique angles
Salazar, Pedro; Rico, Victor; Gonzalez-Elipe, Agustin R.Electrochimica Acta, 235 (2017) 534-542
DOI: 10.1016/j.electacta.2017.03.087
Abstract
In this work we report a non-enzymatic sensor for hydrogen peroxide (H2O2) detection based on nanostructured nickel thin films prepared by physical vapor deposition at oblique angles. Porous thin films deposited on ITO substrates were characterized by X-ray diffraction analysis, scanning electron microcopy (SEMs), X-ray photoelectron spectroscopy (XPS) and electrochemical techniques such as Cyclic Voltammetry (CV) and Constant Potential Amperometry (CPA). The microstructure of the thin films consisted of inclined and separated Ni nanocolumns forming a porous thin layer of about 500 nm thickness. Prior to their use, the films surface was electrochemically modified and the chemical state studied by CV and XPS analysis. These techniques also showed that Ni2+/Ni3+ species were involved in the electrochemical oxidation and detection of H2O2 in alkaline medium. Main analytical parameters such as sensitivity (807 mA M(-1)cm(-2)), limit of detection (3.22 mu M) and linear range (0.011-2.4 mM) were obtained under optimal operation conditions. Sensors depicted an outstanding selectivity and a high stability and they were successfully used to determine H2O2 concentration in commercial antiseptic solutions.
Mayo, 2017 · DOI: 10.1016/j.electacta.2017.03.087
Nanotecnología en Superficies y Plasma - Materiales y Procesos Catalíticos de Interés Ambiental y Energético
Critical Role of Oxygen in Silver-Catalyzed Glaser-Hay Coupling on Ag(100) under Vacuum and in Solution on Ag Particles
Orozco, N; Kyriakou, G; Beaumont, SK; Sanz, JF; Holgado, JP; Taylor, MJ; Espinos, JP; Marquez, AM; Watson, DJ; Gonzalez-Elipe, AR; Lambert, RMACS Catalysis, 7 (2017) 3113-3120
DOI: 10.1021/acscatal.7b00431
Abstract
The essential role of oxygen in enabling heterogeneously catalyzed Glaser–Hay coupling of phenylacetylene on Ag(100) was elucidated by STM, laboratory and synchrotron photoemission, and DFT calculations. In the absence of coadsorbed oxygen, phenylacetylene formed well-ordered dense overlayers which, with increasing temperature, desorbed without reaction. In striking contrast, even at 120 K, the presence of oxygen led to immediate and complete disruption of the organic layer due to abstraction of acetylenic hydrogen with formation of a disordered mixed layer containing immobile adsorbed phenylacetylide. At higher temperatures phenylacetylide underwent Glaser–Hay coupling to form highly ordered domains of diphenyldiacetylene that eventually desorbed without decomposition, leaving the bare metal surface. DFT calculations showed that, while acetylenic H abstraction was otherwise an endothermic process, oxygen adatoms triggered a reaction-initiating exothermic pathway leading to OH(a) + phenylacetylide, consistent with the experimental observations. Moreover, it was found that, with a solution of phenylacetylene in nonane and in the presence of O2, Ag particles catalyzed Glaser–Hay coupling with high selectivity. Rigorous exclusion of oxygen from the reactor strongly suppressed the catalytic reaction. Interestingly, too much oxygen lowers the selectivity toward diphenyldiacetylene. Thus, vacuum studies and theoretical calculations revealed the key role of oxygen in the reaction mechanism, subsequently borne out by catalytic studies with Ag particles that confirmed the presence of oxygen as a necessary and sufficient condition for the coupling reaction to occur. The direct relevance of model studies to a mechanistic understanding of coupling reactions under conditions of practical catalysis was reaffirmed.
Mayo, 2017 · DOI: 10.1021/acscatal.7b00431
Reactividad de Sólidos
Synthesis, Characterisation, and Photocatalytic Behaviour of Mesoporous ZnS Nanoparticles Prepared Using By-Product Templating
Emrooz, HBM; Rahmani, AR; Gotor, FJAustralian Journal of Chemistry, 70 (2017) 1099-1105
DOI: 10.1071/CH17192
Abstract
High surface area mesoporous ZnS nanoparticles (MZN) were obtained with the aid of the by-product of the synthesising reaction. This by-product, namely NaNO3, can be considered as a soft template responsible for the formation of pores. Ethanol and water were chosen as the synthesis media. Ultrasonic waves were used as an accelerator for the synthesis of MZNs. Photocatalytic activities of the synthesised samples for the degradation of methylene blue (MB) were investigated under ultraviolet irradiation. Synthesised specimens were characterised using field emission scanning electron microscopy, transmission electron microscopy, powder X-ray diffraction, diffuse reflectance spectroscopy, N-2-physisorption, and FT-IR spectroscopy. Results indicated that the synthesis media has a pronounced effect on the surface properties of the final porous particles by several mechanisms. The specific surface area of the MZN samples synthesised in water and ethanol were determined to be 53 and 201m(2)g(-1), respectively. The difference in the specific surface area was attributed to the weak solvation of S2- ions (Na(2)S5H(2)O in ethanol) and also to the by-product of the synthesis reaction. The photocatalytic behaviour of the mesoporous ZnS nanoparticles synthesised in these two media were investigated and the results have been interpreted with the aid of effective surface area, pore volume, and bandgap energy of the specimens.
Mayo, 2017 · DOI: 10.1071/CH17192
Materiales Ópticos Multifuncionales
Design and Realization of a Novel Optically Disordered Material: A Demonstration of a Mie Glass
Miranda-Munoz, Jose M.; Lozano, Gabriel; Miguez, HernanAdvanced Optical Materials, 5 (2017) art. 1700025
DOI: 10.1002/adom.201700025
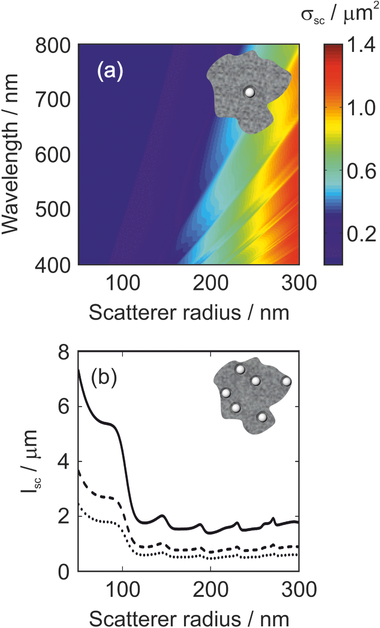
Abstract
Herein, a diffusive material presenting optical disorder is introduced, which represents an example of a Mie glass. Comprising spherical crystalline TiO2 nanoparticles randomly dispersed in a mesoporous TiO2 matrix, it is proved that the scattering of light in this inhomogeneous solid can be predicted in an unprecedented manner from single-particle considerations employing Mie theory. To that aim, a study of the dependence of the key parameters employed is performed to describe light propagation in random media, i.e., the scattering mean free path and the transport mean free path, as a function of the size and concentration of the spherical inclusions based on a comparison between experimental results and analytical calculations. It is also demonstrated that Mie glasses enable enhanced fluorescence intensity due to a combined absorptance enhancement of the excitation light combined with an improved outcoupling of the emitted light. The method offers the possibility to perform a deterministic design for the realization of a light diffuser with tailor-made scattering properties.
Mayo, 2017 · DOI: 10.1002/adom.201700025
Fotocatálisis Heterogénea: Aplicaciones
High UV-photocatalytic activity of ZnO and Ag/ZnO synthesized by a facile method
C. Jaramillo-Páez; J.A. Navío; M.C. Hidalgo; M. MacíasCatalysis Today, 284 (2017) 121-128
DOI: 10.1016/j.cattod.2016.11.021
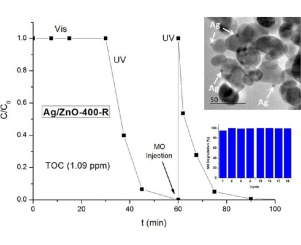
Abstract
ZnO nanoparticles have been successfully synthesized by a facile precipitation procedure by mixing aqueous solutions of Zn(II) acetate and dissolved Na2CO3 at pH ca. 7.0 without template addition. We have investigated the effect of annealing temperature in the final surface and structural properties. Photocatalytic studies were performed using two selected substrates, Methyl Orange and Phenol, both as single model substrates and in mixtures of them.
It has been stated that calcination treatments lead to a significant improvement in the photocatalytic properties of the studied samples, even better than TiO2(P25). As expected, the addition of Ag+ during the photocatalytic degradation of MO increases the reaction rate of the degradation of MO, giving a resultant Ag/ZnO photocatalyst which, after recovery, can be reused at least 18 times for the MO degradation tests, being even more photoactive than ZnO.
Abril, 2017 · DOI: 10.1016/j.cattod.2016.11.021
Reactividad de Sólidos
Effect of Thermal Pretreatment and Nanosilica Addition on Limestone Performance at Calcium-Looping Conditions for Thermochemical Energy Storage of Concentrated Solar Power
Valverde, Jose Manuel; Barea-Lopez, Manuel; Perejon, Antonio; Sanchez-Jimenez, Pedro E.; Perez-Maqueda, Luis A.Enery & Fuels, 31 (2017) 4226-4236
DOI: 10.1021/acs.energyfuels.6b03364
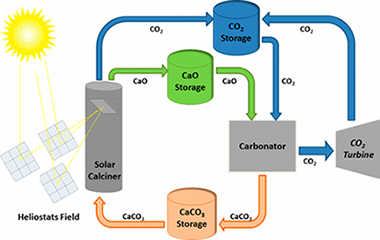
Abstract
The share of renewable energies is growing rapidly, partly in response to the urgent need for mitigating CO2 emissions from fossil fuel power plants. However, cheap and efficient large-scale energy storage technologies are not yet available to allow for a significant penetration of renewable energies into the grid. Recently, a potentially low-cost and efficient thermochemical energy storage (TCES) system has been proposed, based on the integration of the calcium-looping (CaL) process into concentrated solar power plants (CSPs). The CaL process relies on the multicycle carbonation/calcination of CaO, which can be derived from calcination of widely available, cheap, and nontoxic natural limestone (CaCO3). This work explores the effect on the multicycle activity of limestone-derived CaO of thermal pretreatment under diverse atmospheres and the addition of nanosilica, which would be expected to hinder CaO grain sintering. Importantly, optimum CaL conditions for CSP energy storage differ radically from those used in the application of the CaL process for CO2 capture. Thus, calcination should be ideally carried out under low CO2 partial pressure at moderate temperature (below 750 degrees C), whereas CO2 concentration and temperature should be high for carbonation in order to maximize thermoelectric efficiency. When limestone is subjected to carbonation/calcination cycles at these conditions, its performance is critically dependent on the type of pretreatment. Our results indicate that the multicycle CaO activity is correlated with the size of the particles and the CaO pore size distribution. Thus, CaO activity is impaired as particle size is increased and/or CaO pore size is decreased. These observations suggest that pore plugging poses a main limitation to the multicycle performance of limestone-derived CaO at the optimum CaL conditions for TCES in CSPs, which is supported by scanning electron microscopy analysis. Strategies to enhance the performance of natural limestone at these conditions should be therefore oriented toward minimizing pore plugging rather than CaO grain sintering, which stands as the main limitation at CaL conditions for CO2 capture.
Abril, 2017 · DOI: 10.1021/acs.energyfuels.6b03364
- ‹ anterior
- 18 of 37
- siguiente ›















